|
An ion is made up of both positively charged and negatively charged elements. Ions, which are charged atoms and groups of atoms, are found in a wide range of chemical compounds. When sodium and chlorine form an ionic bond, they each have eight valence electrons. When one of the electrons in Na donates one of its electrons to another in a chemical reaction, the resulting positive ion (Na+) and negative ion (Cl) form a stable ionic compound (sodium chloride common table salt). What Is The Ionic Bonding Of Sodium And Chloride? It is the result of their attraction to their opposite charges that sodium and chloride ions form an ionic bond. As a result of this electron transfer, the Na+ and Cl– ionic bonds are formed. What Type Of Bond Is Formed Between Sodium And Chlorine?īecause sodium and chlorine both lose electrons when they come into contact, they have a good chance of bonding. ionic bonding allows us to form all of the molecules that make up our food. Ionic bonding allows us to form substances such as water and table salt, which are needed for survival. Na, a metal, and chloride, a nonmetal, are both made up of ionic bonds, which are composed of sodium (Na).Īs a result of ionic bonding, we have many different substances in our bodies and in our lives. Ionic bonds are typically made up of metal ions and nonmetal ions. Ionic bonds are formed by the electrostatic attraction of ions that are opposite of each other. How Do Sodium And Chlorine Bond Together? Because sodium electrons are held in the Na*Cl ionic bond by chlorine, donating one electron to the chemical element is the only way to get them. How many valence electron pairs are there between sodium and chloride? One valence electron is assigned to sodium, and seven are assigned to chlorine. The positive sodium atom attracts the negative chlorine atom, resulting in the formation of a compound. Each sodium atom in this compound loses one electron (which means it has a charge of one), while each chlorine atom gains one electron (which means it has a charge of one). Do sodium and chlorine lose electrons? NaCl, for example, is a common example of sodium chloride. When a neutral sodium atom reacts with a neutral chlorine atom, the valence electron of the neutral atom is transferred to the chlorine atom, resulting in Cl- and Na+ ions being charged with each other. What occurs when sodium and chlorine are mixed together? These two molecules form an ionic bond because they have a strong electronegativity diference. The Ionic Bond Between Sodium And Chlorine Lauren becomes positively charged when every atom of chlorine gains an electron. Because of this change in electron count, it is possible to find ionic bonds between sodium and chlorine. The valence electron of sodium is one, while the valence electron of chlorine is seven. When sodium and chlorine are combined, they form an electron configuration that is vastly different. The ion has a +1 charge because 11 protons are in the nucleus, but only ten electrons are in the nucleus. Na2 has the same electron configuration as neon (1s2 2s22p6). What Happens To The Valence Electrons Between Sodium And Chlorine?Ĭhlorine neutralizes the valence electron in the sodium atom in order for it to form an ionic bond. If sodium can transfer it’s spare electron to chlorine (as shown in the image above), both atoms will meet their full outer shell requirements and an ionic bond will form. Na and chlorine are frequently at odds because they lose electrons and form an electron with each other, forming a -1 ion. 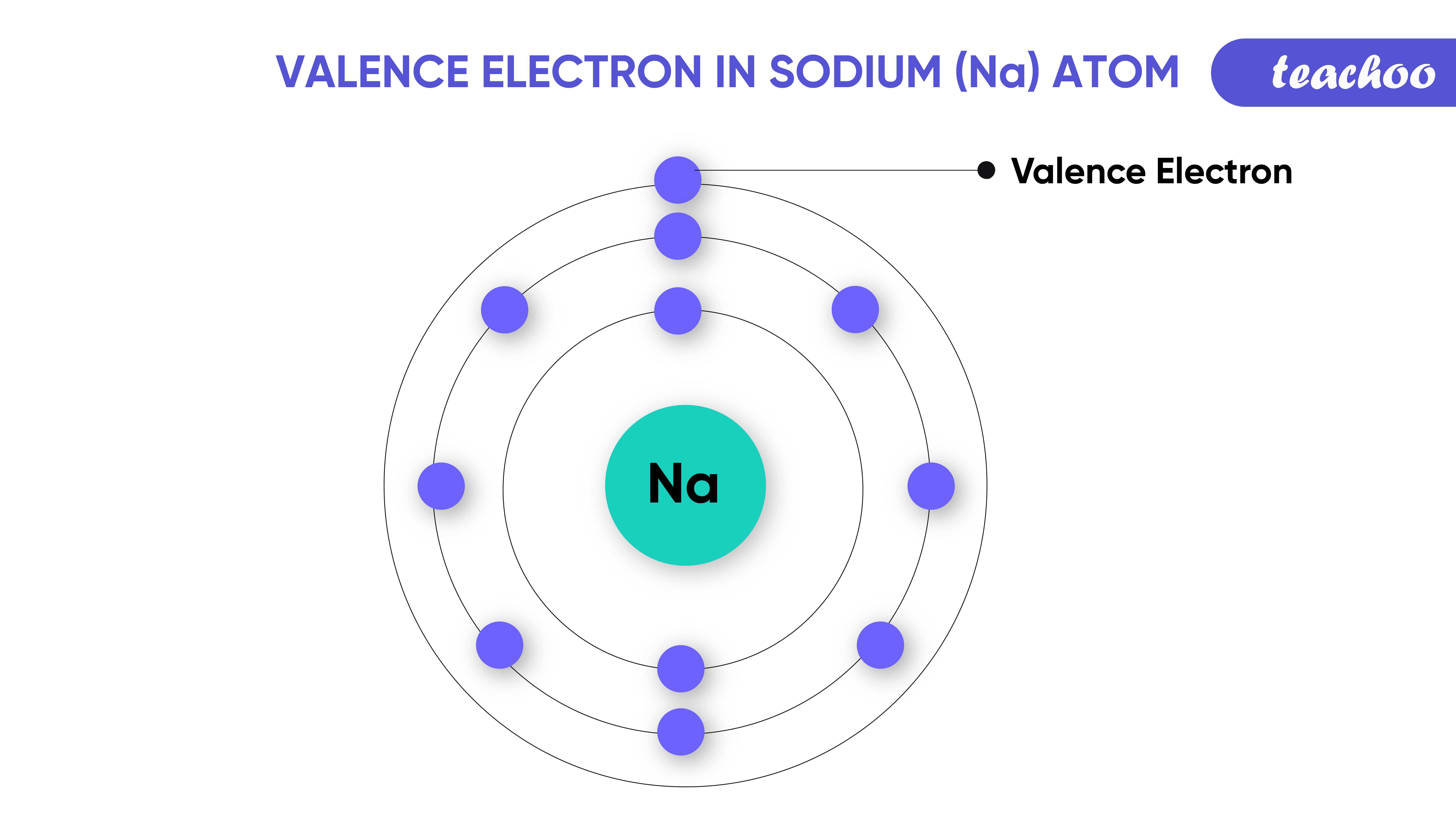
Because they are ionic bonds, they do not share electrons. Only covalent and co-ordinate bonds bind electrons to one another. Na+1), and chlorine requires one electron for stability, they both have the same valency. Because of the fact that sodium has one extra electron in its valence shell (i.e. Metals such as sodium and chlorine are not alloys. We can see that the resulting compound has a net charge of zero. In this case, the sodium cation is balanced by the chlorine anion. The octet of chlorine consists of seven valence electrons, which require one electron more to complete.Īs a result, the sodium atom donates a valence electron to the chlorine atom. Because sodium’s outer shell has only one electron, it has a valency of $ ext -1 $. 
The number of bonds an atom can form to achieve stability, also known as its valency, is considered to be a measure of its stability. When they combine, they form a compound called sodium chloride. This means that they can easily form a bond with another element. One of their similarities is that they both have one valence electron. They are both in the same column, which means they have similar properties. Sodium and chlorine are both elements on the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
